Energy From Nuclear Reaction | How Photovoltaic Cells WorkEnergy From Nuclear ReactionInitially, the energy a photovoltaic cell uses comes from the sun. There, hydrogen nuclei fuse with each other to form helium nuclei and energy. It takes four hydrogen nuclei to form one helium nucleus.
PhotonsPhotons are the energy byproducts of the nuclear reaction in the sun. They are essentially "packets of energy."
Electrons Absorb PhotonsWhen photons from the sun hit a photovoltaic cell, they may be absorbed by an electron. With this extra energy, the electron may become excited and break off its atom, and eventually begin an electric current.
Silicon WaferThe silicon wafer is the basic starting material of photovoltaic cells. As other materials are added to both sides of the cell, the silicon remains neutral, and acts as a barrier layer. This is because of its four valence electrons.
Positive LayerIn a photovoltaic cell, boron is found on the bottom. This forms a positive layer. Often, silicon wafers are obtained with boron already infused into one side.
Negative LayerIn a photovoltaic cell, phosphorus is found on the top, directly on top of the silicon. The phosphorus is a dopant, which forms a negative layer on the side of the cell facing the sun.
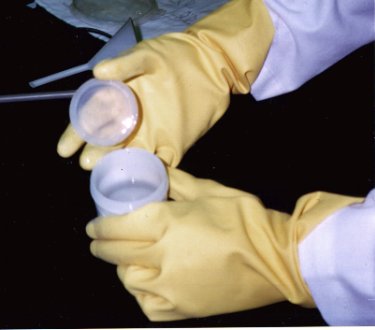
Electrons Move Into PhosphorusThe phosphorus layer, with a negative charge, can produce an electric current when the absorbed charge attempts to "spread out." Shown here is a picture of phosphorus dopant: a clear, odorous fluid.
Photoelectric EffectThe photoelectric effect explains the movement of electrons in the presence of photons. This was first hypothesized in 1839, but was not fully explained until 1921, when Albert Einstein won the Nobel Prize for it.
|